
Note the non-linear shell ordering, which comes about due to the different energies of smaller and larger shells.The construction of the periodic table ignores these irregularities and is based on ideal electron configurations.
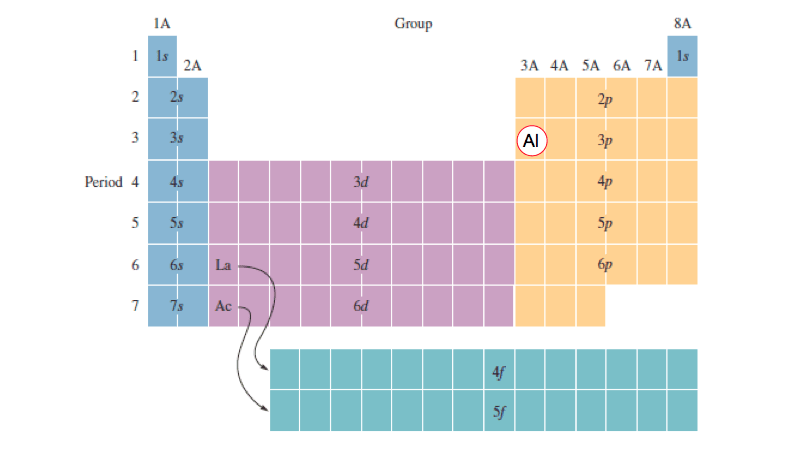
In many cases, multiple configurations are within a small range of energies and the small irregularities that arise in the d- and f-blocks are quite irrelevant chemically. 3 valence electrons, all have electron configurations terminating in ns2.

